Research
Application of reversible ionic liquids in reactions of steroids
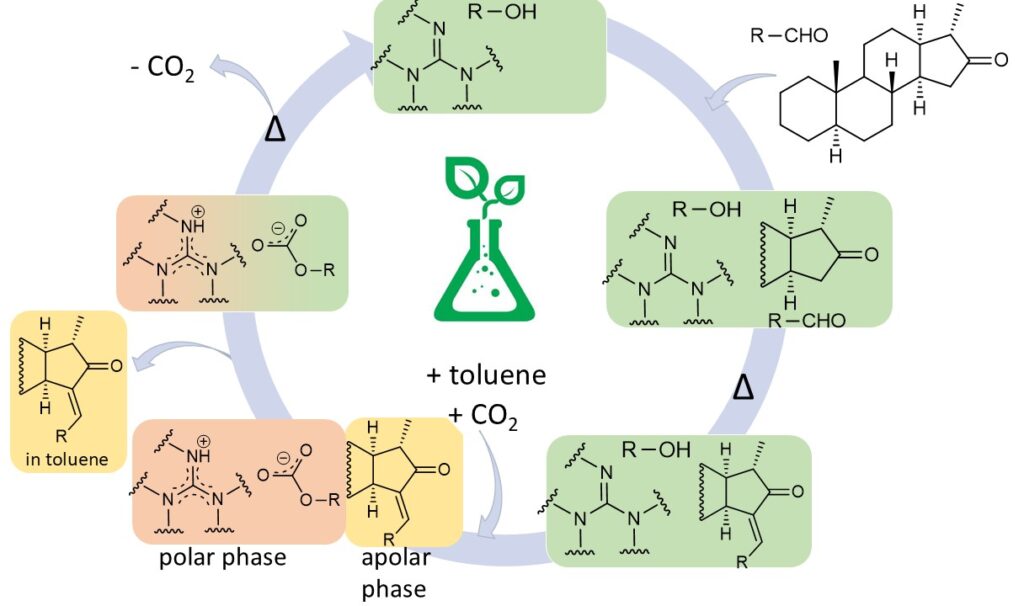
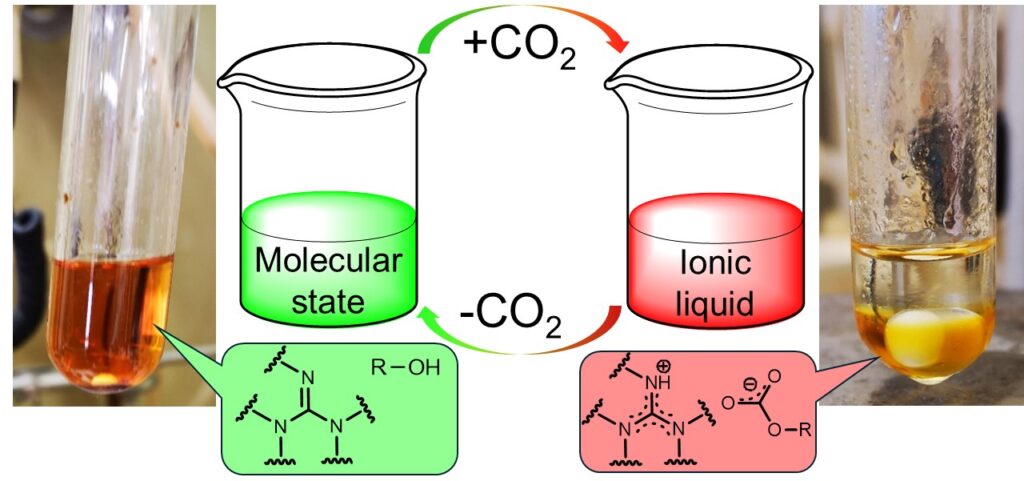
Reversivle ionic liquids (or switchable polarity solvens) are capable of changing their polarity reversibly between a nonionic and the ionic form in the presence of an external trigger (e.g. carbon dioxide). The base of this system are liquid bases (in our work a guanidine derivatives) and alcohols, which could be used as a catalyst and a solvent as well.
Read more
Benzylidene derivatives obtained by the Claisen-Schmidt condensation of steroidal ketones may serve as starting materials for the synthesis of compounds of pharmacologival importance or may show favourable biological acivity themselves. The 16-arylidene group was found to be a good pharmacophore for citotoxic acivity.
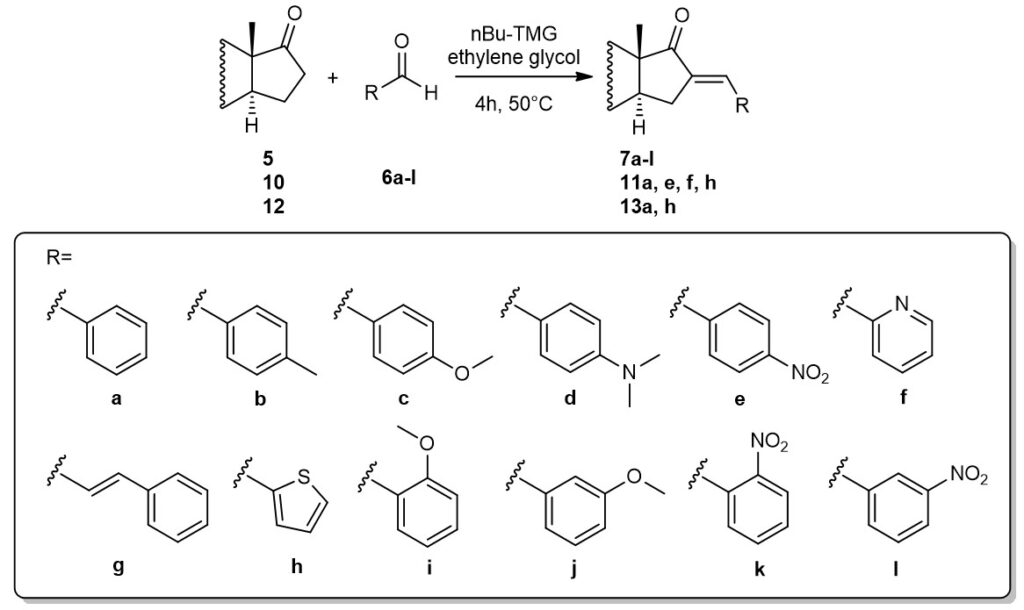
Claisen-Schmidt condensation of aromatic aldehydes and different 17-oxo-steroids with androstane and estrone skeleton was carried out.
The mixture of nBu-TMG (1,1,3,3-tetramethyl-guanidine) and ethylene glycol, used as a catalyst and solvent as well, were found to be reusable in the synthesis of steroidal products without significant decrease in the yields.
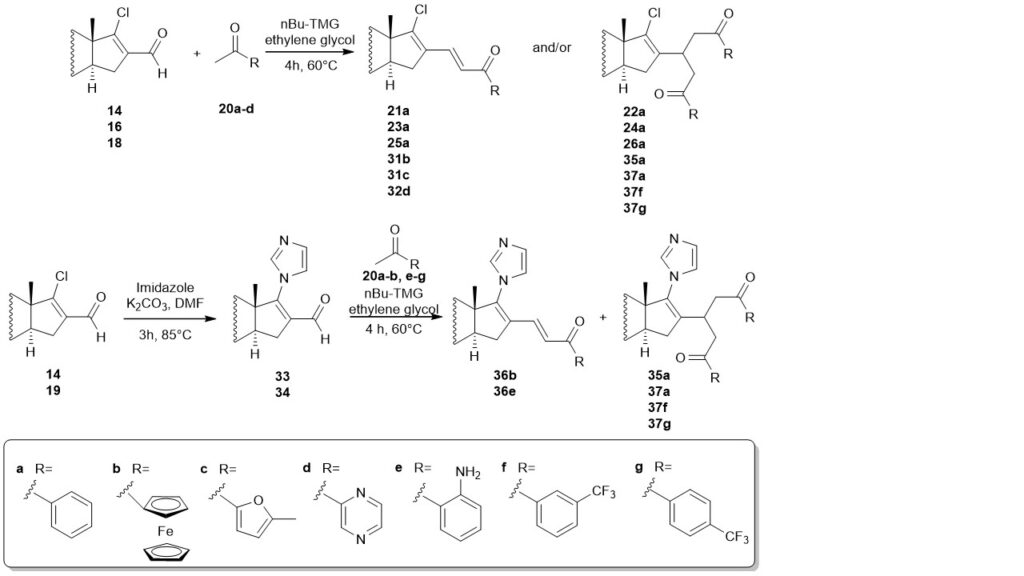
Different 17-chloro-16-formyl steroids (14, 16, 18) and 16-formyl-17-imidazolyl-steroids (33, 34) with androstane and estrone skeleton were synthesized and their Claisen-Schmidt condensation with aromatic ketones (20a-g) was performed. Among the 18 compounds synthesized, 14 steroids were new.
It was established that a subsequent Michael addition can also take place in addition to the Claisen-Schmidt condensation. The ratio of the two products could be controlled well by the reaction conditions. Guanidines with higher basicity provided the products with good yield even at lower temperatures, but the ionic liquids, formed from them with carbon dioxide, could not be decomposed. The nBu-TMG/ethylene glycol mixture was an effective catalyst that can be reused three times to obtain the products (21a, 35a) without a decrease in the yield. 15 steroids with a new structure were synthesized by the use of the method.
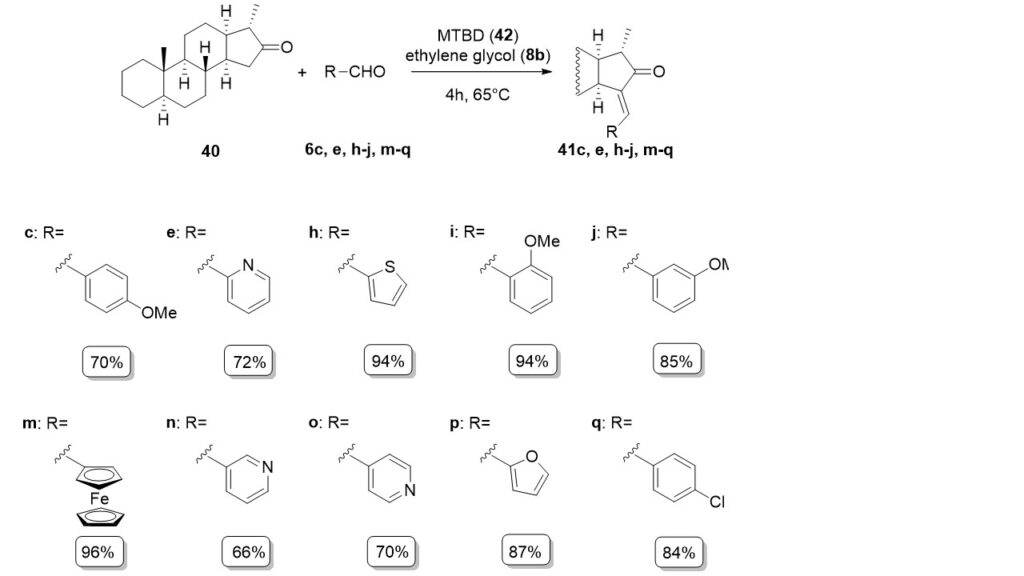
15-Arylidene steroids were obtained via the Claisen-Schmidt condensation between an unnatural 16-oxo-18-nor-5α-13α-steroid (40) and aromatic aldehydes in the presence of MTBD (7-methyl-1,5,7-triazabicyclo[4.4.0]dec-5-ene) (42) and ethylene glycol.
During this work 11 new steroid compounds were synthesized with good yields. The ionic liquid, which was formed from MTBD, ethylene glycol and carbon dioxide, has not been reported in the literature. By using this method compound 41a was synthesized in excellent yield through five cycles.
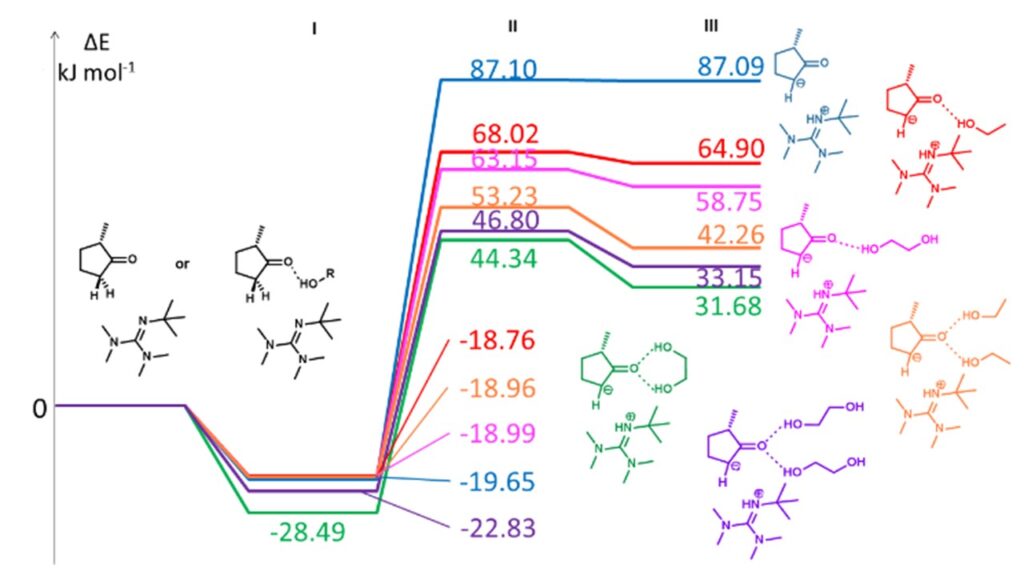
It was found that 1,2- (or 1,3-) diols have a key role in the Claisen-Schmidt reaction: they facilitate the splitting of the CH-acidic hydrogen, which is the rate-determining step of the reaction, by forming two hydrogen bonds with the 16-oxo group., So ethylene glycol participates in the reaction not only as a co-solvent, but also as a co-catalyst.
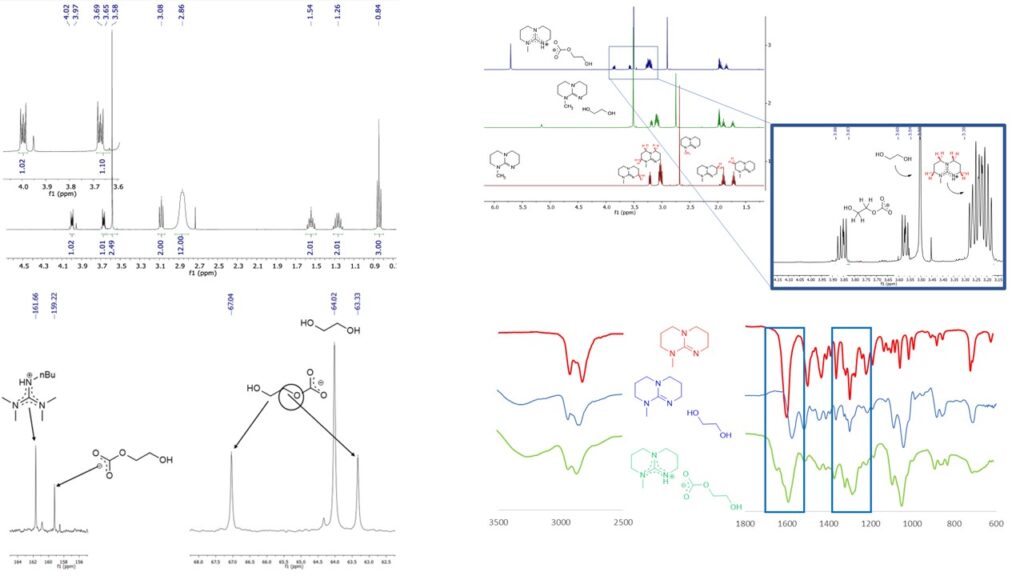
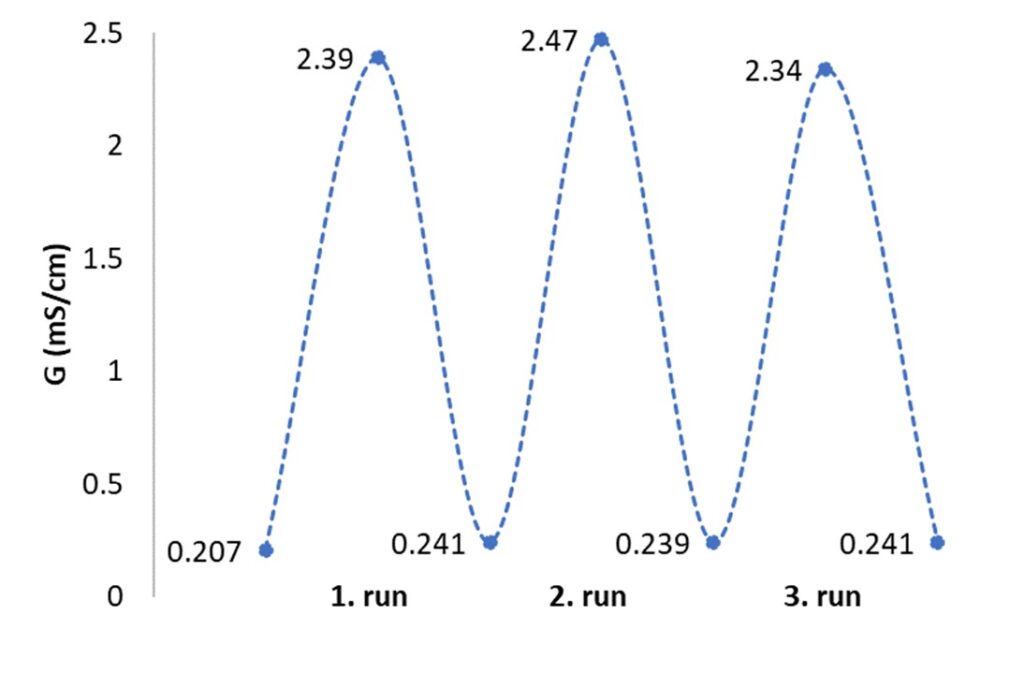
The structure of the new ionic liquids was determined. It was established by using IR and NMR measurements that only one of the hydroxyl groups of ethylene glycol was converted to carbonate when the base and alcohol were mixed in a 1/1 molar ratio. The reversibility of the ionic liquid was proved by conductivity measurements.
The mixtures of nBu-TMG/ethylene glycol and MTBD/ethylene glycol were efficiently used for synthesis of steroid derivatives.
Novel chiral aminoalkyl-phosphine ligands
There are several methods for the preparing of enantiomerically pure compounds. Amongst these possibilities, transition metal catalyzed asymmetric synthesis can be mentioned as one of the most powerful methodologies. Chiral bidentate ligands with C1 symmetry represent a unique type of stereselectors due to thier ability to desymmetrize the transition metal complex both sterically and electronically. Chiral phosphine-amin (P,N) type compounds contains belong to a more specific class of C1 symmetry heterodonor ligands. The introduction of a stereogenic nitrogen in a phosphine-amine ligands may further incerase the catalytic efficiency of a transition metal catalyst.
Read more
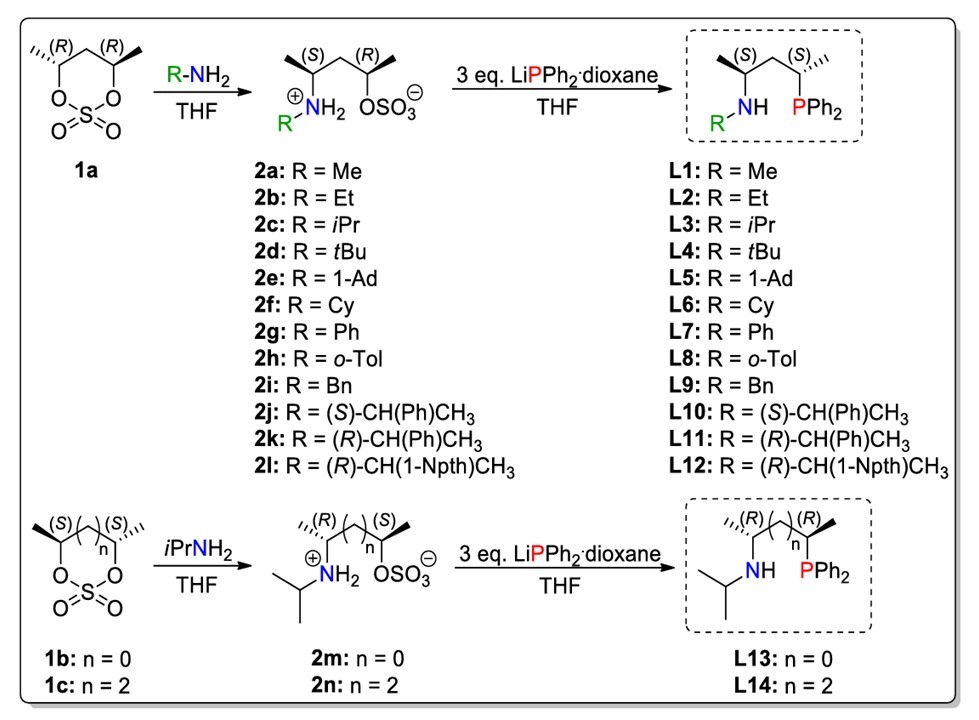
Ten novel (L2, L5-L8,L10-L14) aminoalkyl-phosphine (P,N) type ligans with stereogenic nitrogen was prepared. The synthesis of these compounds was carried out by a simple two-step method enables the preparation of versatile P,N type compounds, that differ in in the nitrogen substituens (L1-L12) and the lenght of P,N backbone (L13 and L14). The structure os the new compounds was investigated by nuclear magnetic resonance and mass spectroscopic methods.
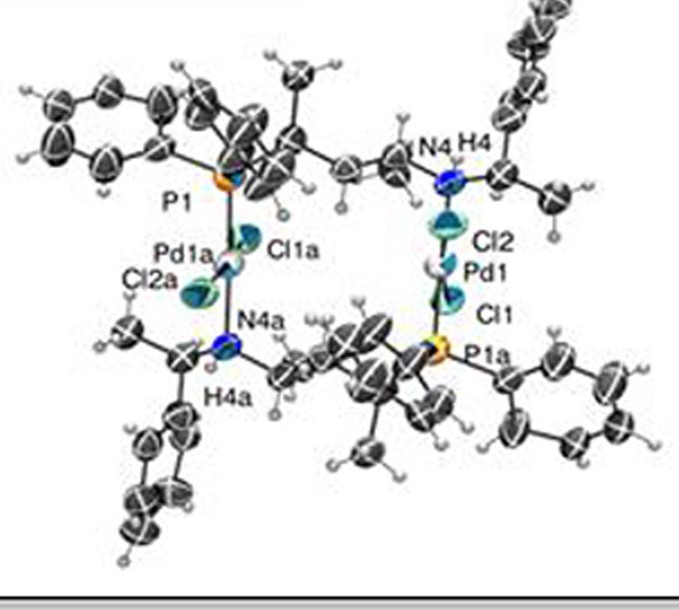
It was estabilished that [Pd(L)Cl2)complexes of pentane-2,4-diyl based ligands L1, L2, L3, L4 and L10 form six-member chelate rings with chair conformation. It has been recognized that the coordination of ligand L1 and L2 having sterically less demanding N-methyl and N-ethyl froup, respectively, results in the formation of an isomeric mixture. In constrast to these, the coordination of ligands L3, L9 and L10 with bulkier iPr-, Bz- and (S)-phenyl-ethyl substituents, respectively, occuors stereoselectively with respect to the configuration of the nitrogen and the conformation of the chelate ring. It has been proven that changing the length of alkane-diyl backbone also leads to the formation of ismoerically realted palladium species. In the case of ligand L13 with butane-2,3-diyl backbone the two five-membered chelates have opposite nitrogen configuration, the cooridnation of hexane-2,4-diyl based ligand L14 results in the formation od multiple complex species.
Ferrocene derivatives as a potential molecular receptors
The numerous applications of ferrocene are due to the interesting chemical and electrochemical properties of the compound as well as its high stability. Its well-defined single-electron reversible oxidation reaction can be used to study various physical and chemical interactions, and its reactivity in electrophilic substitution reactions allows for several structural modification, making it an ideal sensing element for electrochemical sensors. Most of these sensors typically have a binding site that can selectively interact with the analyte to be tested and the resulting interaction can cause a change in the electrochemical behavior of the ferrocene incorporated into the molecule.
Read more
In addition to a previously know compound (5a), a new derivative (5b) was also synthesized by the new method. The palladium-caatalyzed carbonylative Sonogashira reaction, which was earlier used for the synthesis of the corresponding alkynyk ketone (2) intermediate was replaced by a cheaper andd less cumbersome aldol-type addition reaction to give an alkenyl ketone (7) with an analogous structure. During the preparation of 2-amino-6-phenyl-4-ferrocenylpyrimidine, the previously know procedure was modified by the addition of NaOH and the use of THF to avoid the formation of undesired by-products.
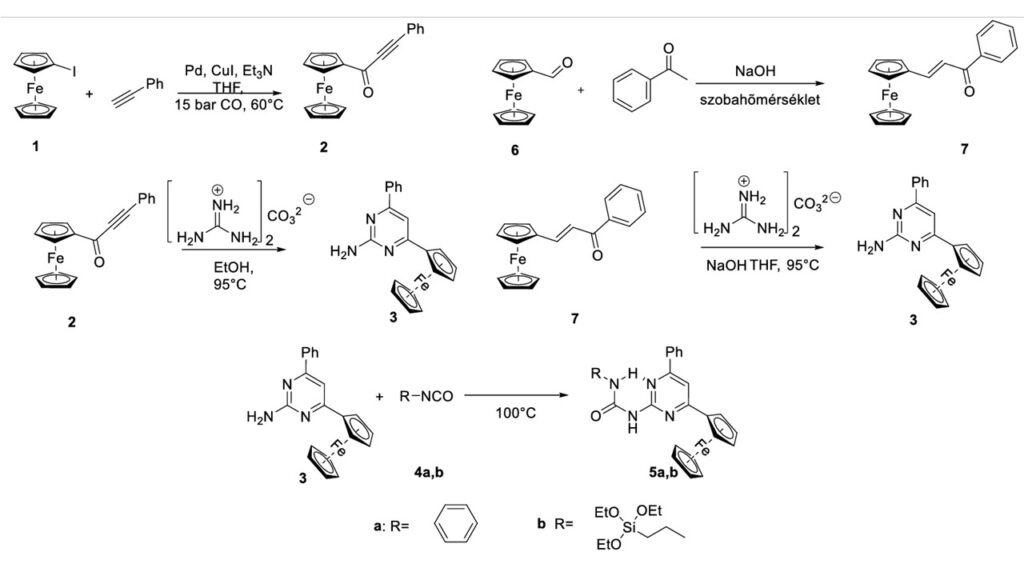
It was found, that the chloroform solution of 5a and 5b compounds contained two different ismoers (5a and 5a‘, as well as 5b and 5b’, similary to other 2-ureido-4-ferrocenyl pyrimidines, examined before). The activation parameters of the 5a-5a’ isomerisation reaction were determined by experimental, as well as by theoretical methods.
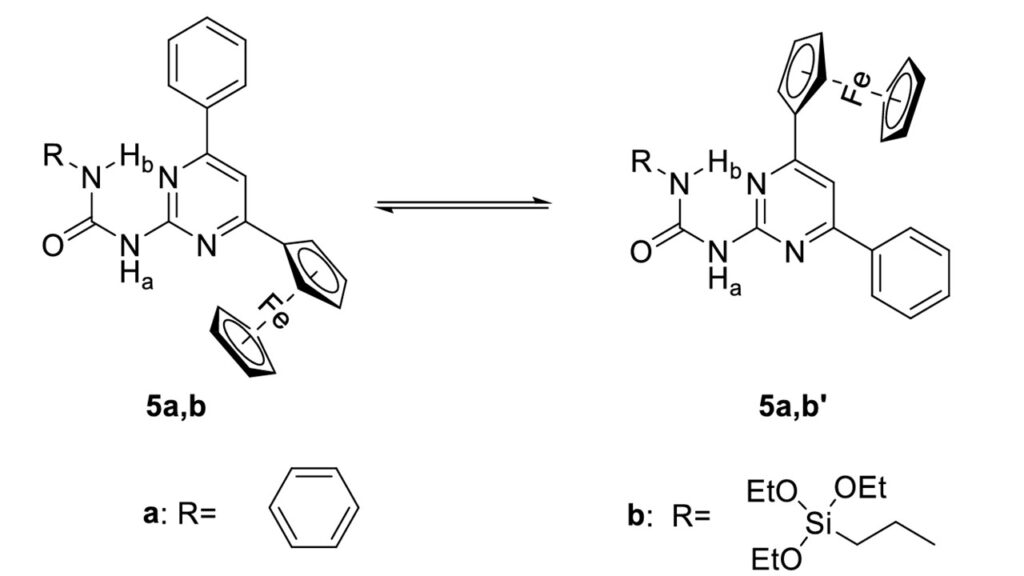
Sol-gel techniques were applied to produce novel ferrocene-containing organic-inorganic hybrid materials. By changing synthetic parameters, the formation of eiter hollow or solid spherical silica particles could be achieved.
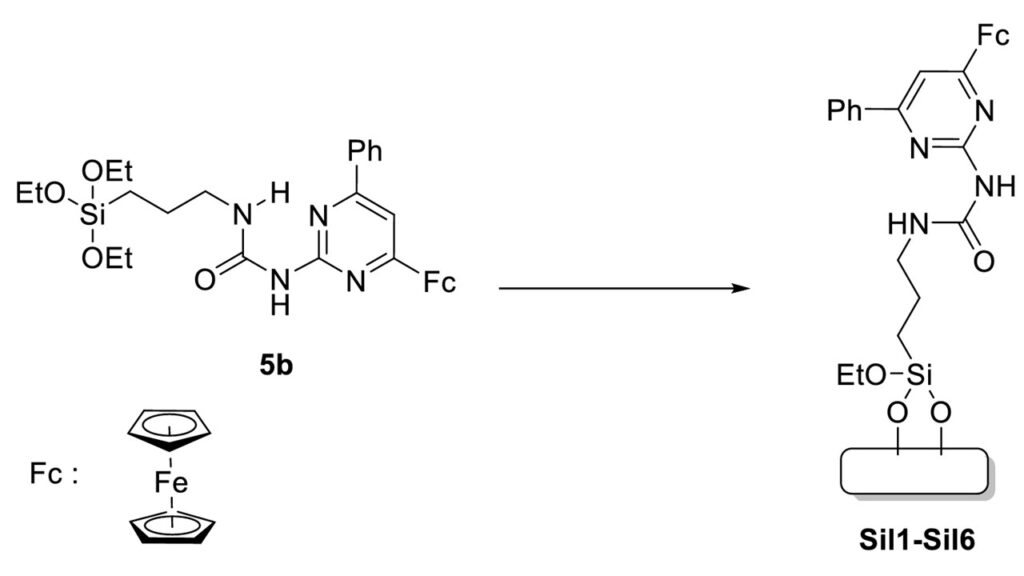
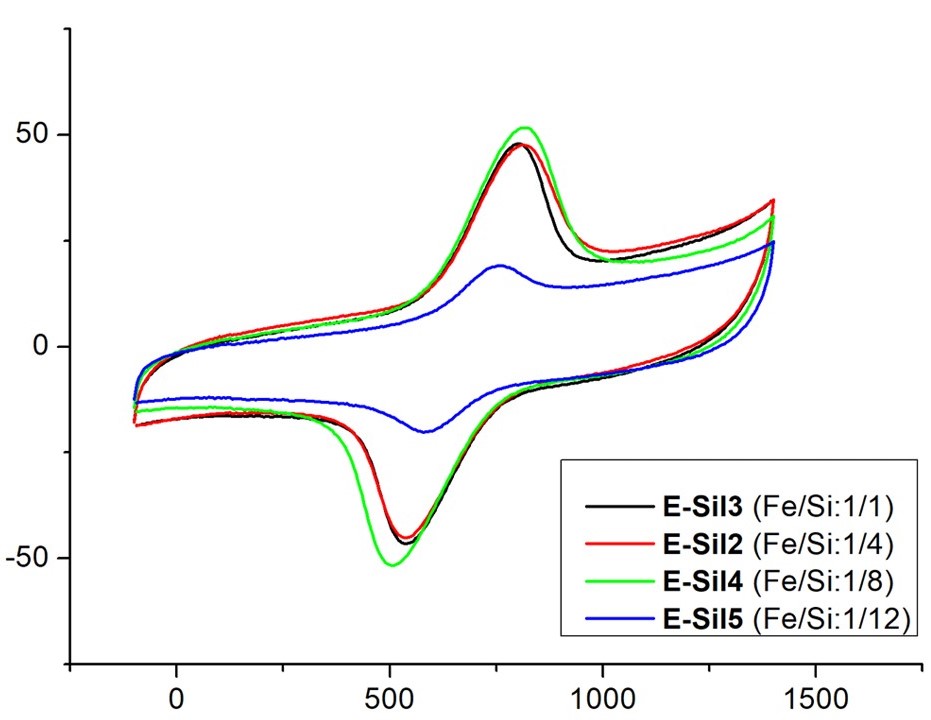
Sol-gel electrodeposition technique was used to obtain ferrocene-containing silica films on surface of spectral graphite electrodes. It was found that the reduction of the Fe/Si ratio led to the decrease of the functionalized silica film on the surface was proved by cyclic voltammetry experiments and SEM studies.
Cyclic voltammetry experiments were applied to verfy the presence of complexes, formed by compound 5b, immobilized on the electrode surface and guest molecules, such as melamine and 2,6-diaminopyridine.
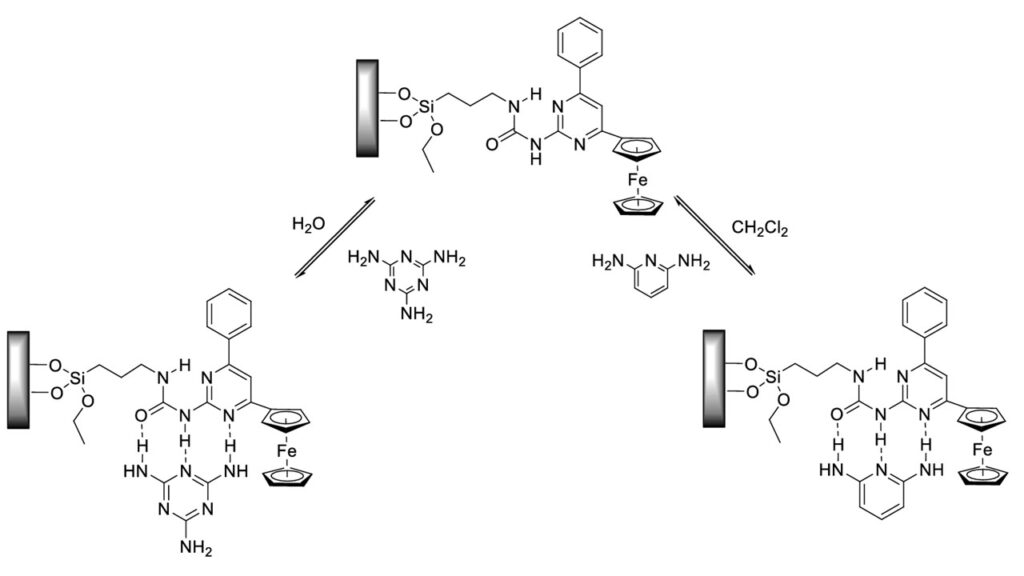

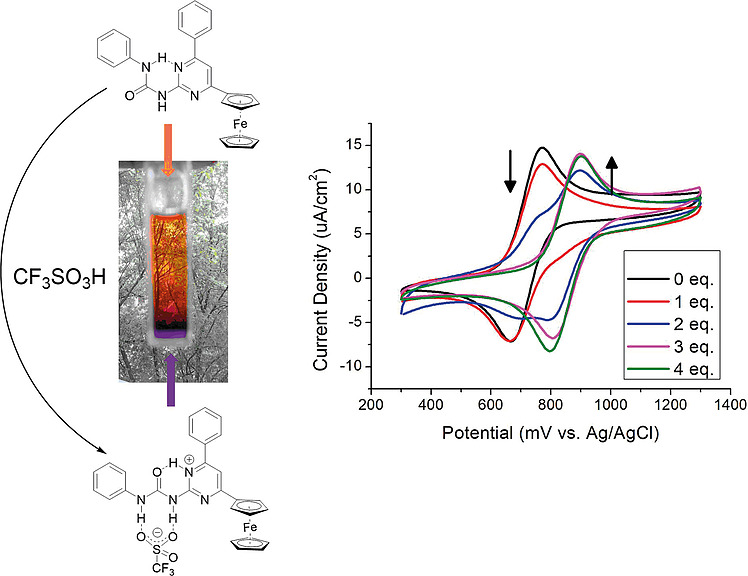
It was demonstrated that the changes detected in UV/Vis and 1H NMR spectra as well as in cyclic voltammograms in the presence of strong acids were results of the protonation and complexation reaction, involving 1-(6-phenyl-4-ferrocenylpyrimidin-2-yl)-3-phenylurea (5a). The molecule undergoes a structural change that allows the binding of anions of the strong acids applied. The structure of the complex formed by compound 5a with trifluoromethanesulfonic acid was also confirmed by X-ray diffraction.
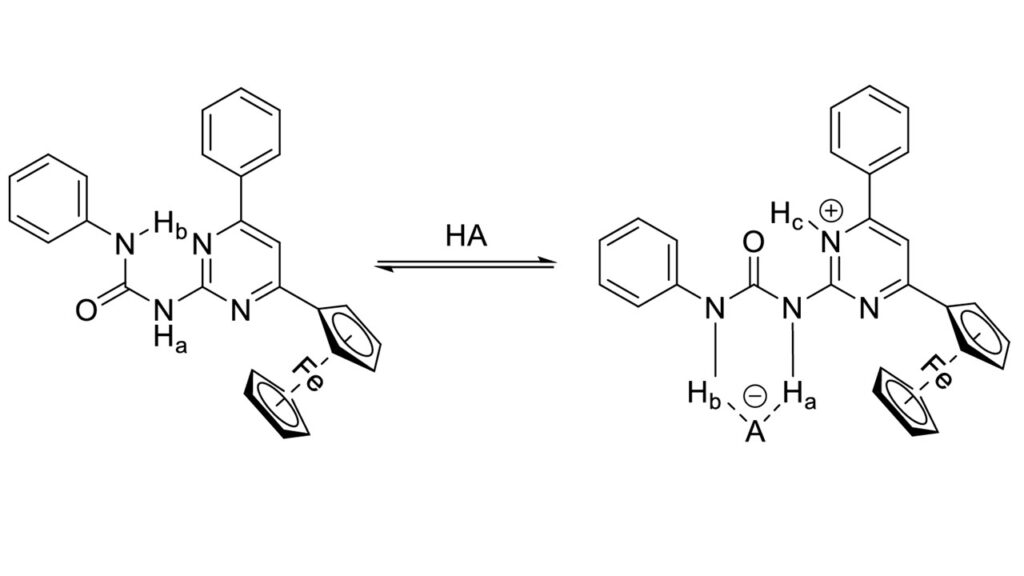

Using quantum chemical calculations, it was showed the relationship between the acid strength of the applied acid and the geometry of the resulting complexes. The results show that the structure of the complexes changes systematically with acid strength. When the acid is weak enough, it is not the 5aH+ ion but the 5a-acid complex that is formed.
Palladium-catalyzed coupling and carbonylation reaction
Palladium catalysts are powerful tools in organic chemistry, catalyzing a variety of reactions and providing exceptional opportunities for the formation of carbon-carbon bonds. The unique properties and outstanding catalytic activity of the metal have driven researchers to develop more active and more efficient catalysts. The development of reusable catalysts as well as minimization of metal loss in order to recycle the expensive metal and meet the demands of the pharmaceutical industry present significant challenges. Heterogeneous catalysts can meet these expectations
Researchers
Dr. Rita Skoda-Földes
Read more
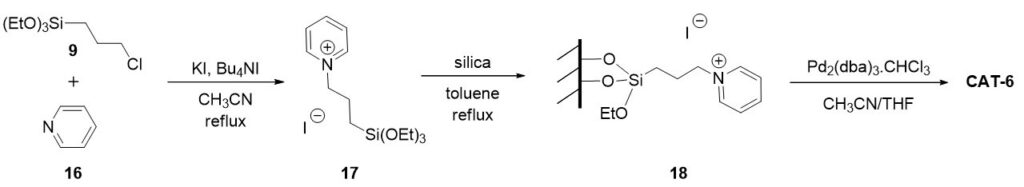
Three heterogeneous palladium catalysts containing adsorbed ionic liquid phase were prepared and their activity and stability were studied in aminocarbonylation reactions. In the presence of the catalyst obtained from the support modified with pyridinium cations excellent activity and selectivity were observed during catalyst recirculation. The dissolution of the ionic liquid was confirmed, which had caused significant palladium leaching.
In order to improve the stability of the catalysts, three heterogeneous catalysts were prepared by using supports with grafted cations. Compared to imidazolium and phosphonium type SILPs, outstanding activity and selectivity as well as good recyclability could be achieved using SILP phases modified by pyridinium cations. Under optimised conditions CAT-6 provided minimal palladium leaching (0.2% in the first run and under 0.13% in further runs).
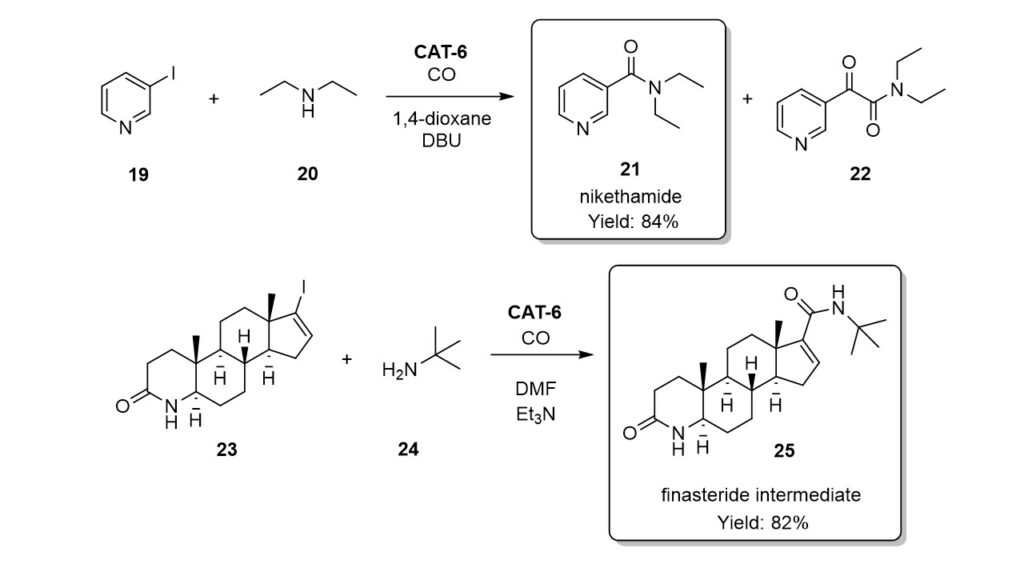
CAT-6 catalyst was succesfully applied in the synthesis of two pharmacologically active derivatives. Excellent selectivity (over 95%) and yield (84%) were achieved in the sythesis of nikethamide. The finasteride intermediate was isolated in 82% yield. The CAT-6 catalyst proved to be reusable in five successive runs in both cases.
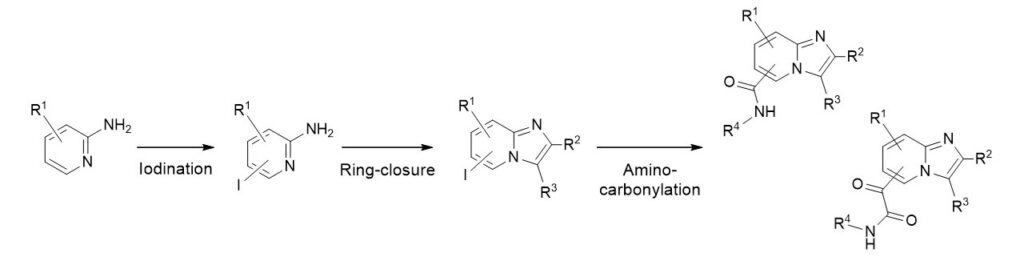
A three-step reaction route was developed to synthesize and functionalize different imidazo[1,2-a]pyridine derivatives.
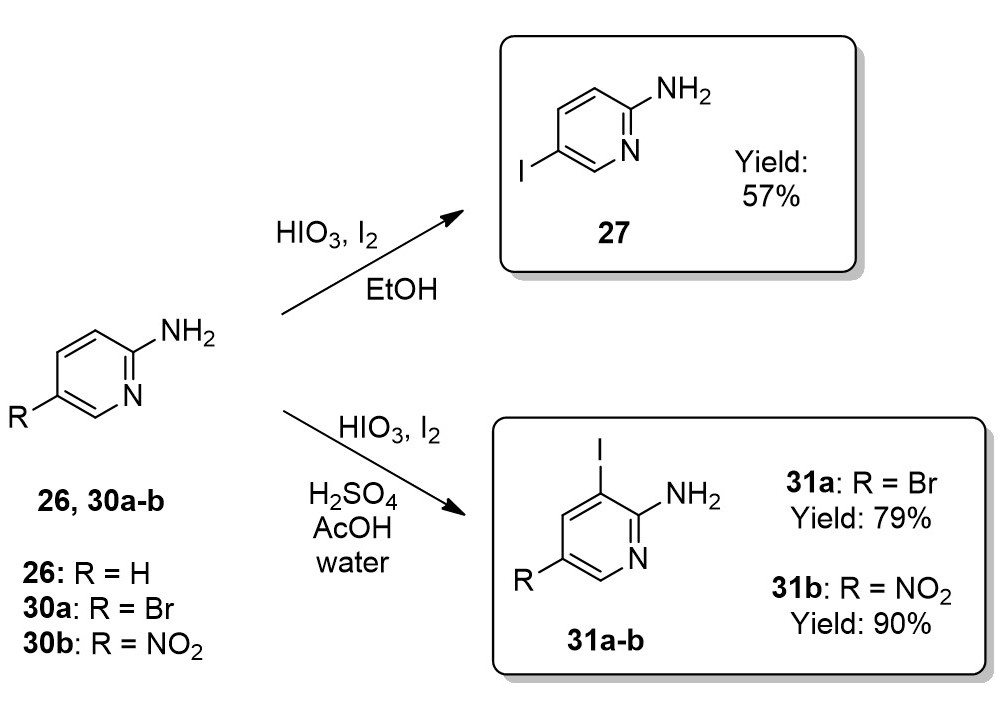
The iodination of 2-aminopyridine derivatives resulted in 2-amino-5-iodopyridine (27), 2-amino-5-bromo-3-iodopyridine (31a) and 2-amino-3-iodo-5-nitropyridine (31b).
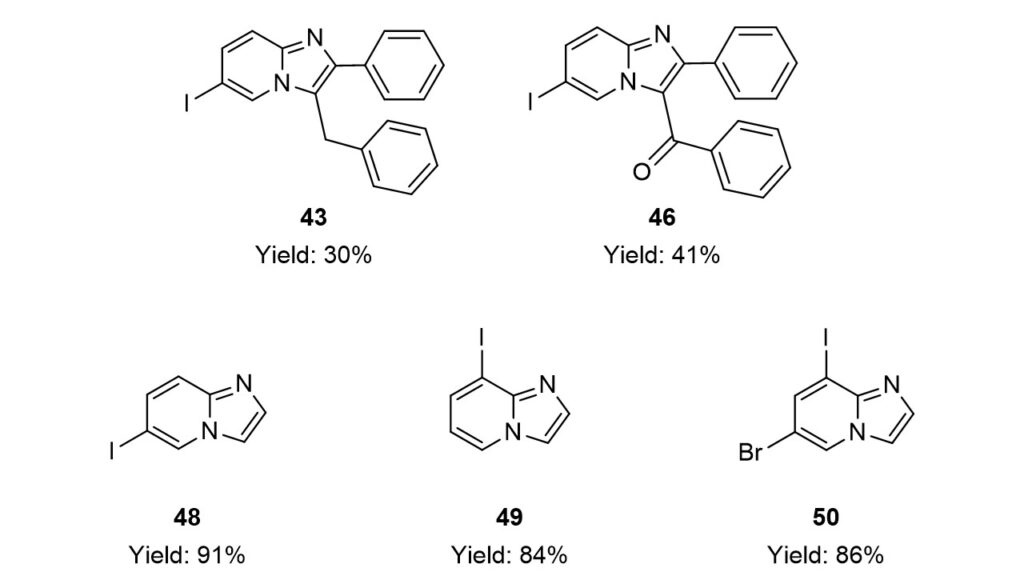
Seven methods were tested to form the imidazo[1,2-a]pyridine core. While the ring closure was easily accomplished under various conditions in the presence of 2-aminopyridines without the iodo substituent, the transformation of iodinated analogs proved to be more challenging. Utilizing the previously gained experience two iodoimidazo[1,2-a]pyridine derivatives (43, 46) were obtained in poor and three (48, 49, 50) in good yields.
The aminocarbonylation reactions of the iodoimidazo[1,2-a]pyridine derivatives were studied in the presence of the heterogeneous catalyst (CAT-6): the effect of the solvent, base, pressure and temperature was investigated. Excellent recyclability of CAT-6 could be achieved using lower temperature and higher pressure (100 °C, 30 bar) together with DMF as solvent and triethylamine as base. Under these conditions dicarbonylation proved to be the favorable reaction. In the presence of toluene/DBU solvent/base system decreased temperature and increased pressure resulted in a selectivity change towards the amide. As it was expected, when aromatic amines were applied as reagents the formation of only monocarbonylated products was observed. Furthermore, aminocarbonylation of 8-iodoimidazo[1,2-a]pyridines showed definite selectivity towards amide products under various conditions even in the case of aliphatic amines. Altogether 17 aminocarbonylated derivatives – 12 amides and 5 ketoamides – were produced in good yields (64-91%).
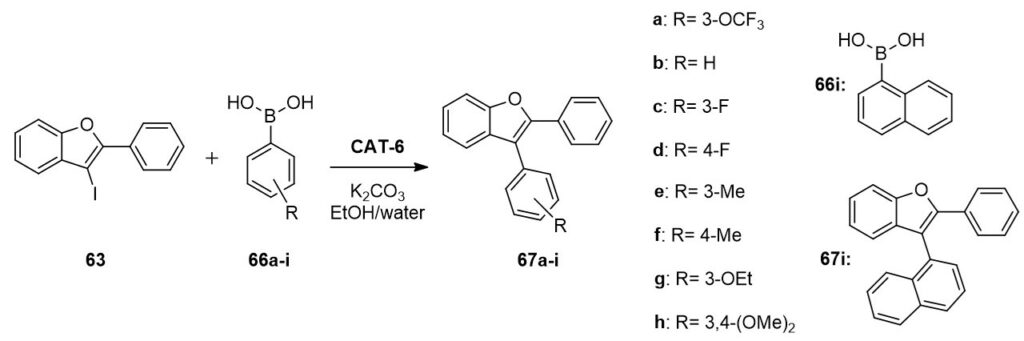
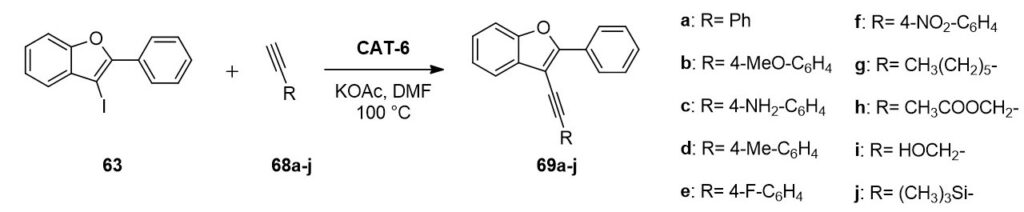
The heterogeneous catalyst (CAT-6) was suitable for the functionalization of 3-iodo-2-phenylbenzo[b]furan (63) in the presence of simple amines, acetylenes and boronic acids, in other words, in addition to aminocarbonylation the applicability of CAT-6 was proven in Suzuki- and Sonogashira coupling as well.
High conversion and yield were achieved in the reaction of 3-iodo-2-phenylbenzo[b]furan (63) and propargylamine in ten consecutive runs.
The use of ethanol/water solvent mixture together with potassium carbonate base was identified as the optimal condition for Suzuki reaction. Under these conditions the catalyst was efficient in at least seven runs without any loss of its activity, moreover the palladium loss was also minimal (0.1-0.9%). This methodology was applied successfully in the synthesis of nine different 3-arylbenzo[b]furan derivatives in 80-95% yields.
In the case of Sonogashira reaction the application of potassium acetate base and a temperature of 100 °C was found to lead to promising results. CAT-6 catalyst was tested both in the presence and in the absence of CuI cocatalyst. Although longer reaction time was required to achieve complete conversion under copper-free conditions, it was still more beneficial with respect to the stability, recyclability of the catalyst and palladium leaching (1-2%) indeed. Ten different 3-alkynylbenzo[b]furan derivatives were synthesized in Sonogashira reaction in 50-92% yields.
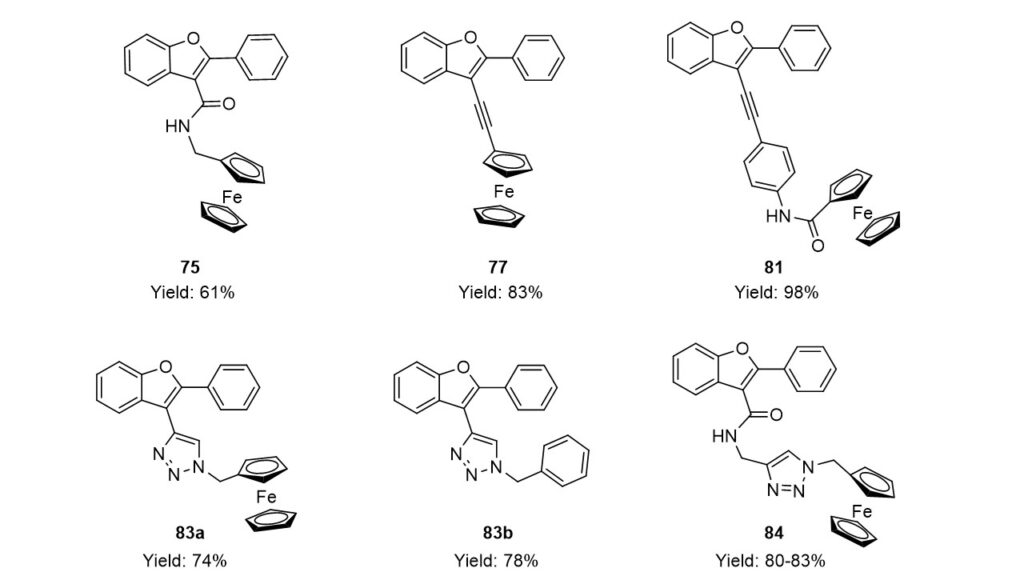
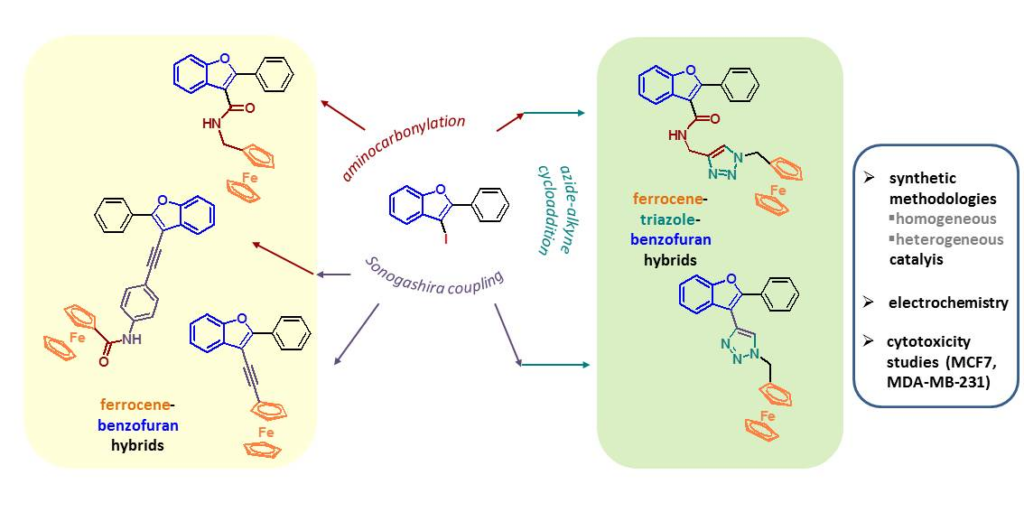
CAT-6 catalyst was found to be efficient in the synthesis of hybrid molecules as well. Three hybrids (75, 77, 81) were successfully prepared by the combination of benzo[b]furan core and ferrocenyl group. Palladium catalyzed reactions followed by azide-alkyne cycloaddition provided one triazole-benzo[b]furan (83b) and two ferrocene-triazole-benzo[b]furan hybrids (83a, 84). The toxicity of ferrocene-benzofuran derivatives on human breast cancer cell lines (MCF7, MDA-MB-231) were also studied. The results led to the conclusion that, in addition to the ferrocenyl group amide and triazolyl groups also play a significant role in the biological effect.